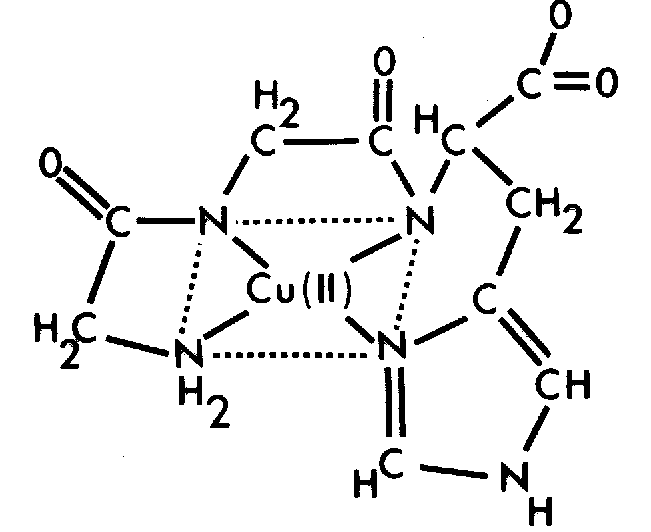
Preclinical research examining GHK-Cu has reported observations involving connective tissue remodelling, collagen organisation, and cellular repair responses in laboratory models. Scientific studies have investigated the peptide’s influence on fibroblast activity, extracellular matrix signalling, and vascular stability within controlled research environments. These findings have contributed to ongoing research exploring the role of copper-binding peptides in biological repair signalling networks and regenerative biology investigations.
Despite these experimental observations, the majority of available research evidence concerning GHK-Cu remains derived from laboratory and animal studies. Controlled human clinical trials evaluating the peptide remain limited, and regulatory authorities have not approved GHK-Cu as a therapeutic compound. Consequently, the peptide continues to be classified as an investigational research compound studied for its biological signalling properties and experimental applications within scientific research environments.